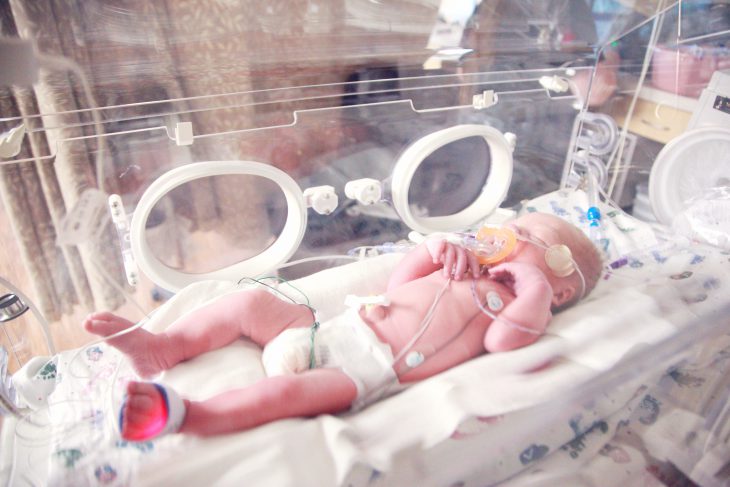
One of the most important systems in our bodies is the immune system. It defends our bodies from germs, infections, bacteria, viruses, and more. But, would you have imagined that about 1 in 58,000 babies are born with little to no Immune system? This is a very rare genetic disorder called Severe Combined Immunodeficiency (SCID). A baby can be diagnosed with SCID before birth through screenings, but most babies are diagnosed with SCID within the first six months of life.
When a child has SCID, it means they lack T cells, Natural Killer Cells, and Functional B cells. T Cells help protect the body from infection and help fight cancer. Natural Killer cells destroy cells infected with a virus. Functional B cells produce antibodies to fight bacteria and viruses.
The process of treating or curing a disease by altering a person’s genes is known as gene therapy. In two previous studies conducted, SCID gene therapy consisted of two generations, both using viruses to deliver the genes.The first generation of treatment worked, but patients unfortunately developed leukemia, a cancer of the white blood cells that makes them abnormally large. The research community did deliver a “second generation” of gene therapies that were safer, but they did not completely restore the immune system.
To treat infants with SCID, stem cells are taken from the bone marrow of siblings, parents, or unrelated donors. Then, a bone marrow transplant introduces these healthy infection-fighting cells into the SCID infant’s body. The idea is that this will provide a new immune system for the patient. Generally, bone marrow stem cell transplants from family donors are effective but unavailable for more than 80% of patients in the world. This means a higher risk of the non-family donor’s T cells attacking and damaging the patient’s healthy cells.
In this study, the researchers used a type of gene therapy involving a “lentiviral vector.” A lentiviral vector is a type of virus called a lentivirus that inserts its RNA into the host’s cells. They took advantage of this virus’s action to insert a corrected gene sequence, as an RNA strand, into the patient’s own bone marrow stem cells to both “fix” the genetic error and reduce the chance of rejection. The gene of interest is IL2RG, which instructs the body to make certain immune proteins in the bone marrow. When this gene is broken, SCID results.
The research team had successfully tried this type of treatment before in children and young adults with SCID. They combined the lentiviral vector gene therapy with a chemotherapy agent called nonmyeloablative busulfan, typically given to patients before a stem cell transplant. This drug destroys a patient’s bone marrow cells in preparation for new stem cells. The researchers hypothesized that lentiviral gene therapy, after a low dose of this drug busulfan, would be a safe and effective treatment for infants with recently diagnosed SCID.
First, the infant’s bone marrow was collected. The correct gene was inserted into the patient’s blood stem cells using the lentivirus “vector” or carrier. The cells were then frozen and went through quality testing in order to detect, reduce, and correct any problems that may have occurred. Importantly, the lentiviral vector contained protectors that prevented the gene therapy from accidentally causing leukemia. The protectors work by blocking the virus from turning on certain “oncogenes” (or cancer-linked genes) that happen to sit next to the IL2RG gene on the chromosome.
They recruited a group of eight infants newly diagnosed with SCID. The researchers conducted their experiment by giving the infants one to two daily doses of busulfan by injection. They customized the initial dose based on the weight and age of the patient and previous knowledge on how this drug typically moves through the body.
The results of the experiment supported the research team’s prediction. Natural Killer cells were restored within the first four months in seven of the eights infants as the T cells. The eighth infant initially developed a low T cell count but improved after a boost of gene-corrected cells without needing additional busulfan pre-treatment. Several types of blood cells such as T, B and natural killer cells made in the bone marrow seemed active within 3-4 months after infusion with the viral lentiviral vector.
The combination of lentiviral gene therapy with busulfan conditioning appeared safe in all eight infants. These results aligned with what the researchers expected. Patients were followed for a range of 6-24 months after the study to assess whether their new immune system remained stable. After the 24 month period, they concluded that this treatment was more effective than current treatments for SCID patients with fewer side effects.